Yashita Chaudhary, Grade 12
In the case of a crisis, this is a recurring issue and is seen more frequently on a smaller scale, when individual patients struggling with life-threatening diseases, battle using unapproved medical products. To alleviate these problems, there needs to be a greater focus on supportive care, repurposed drugs, and expanded access.
In the book, Integrating Clinical Research into Epidemic Response: The Ebola Experience, there is an emphasis on the urgency of early supportive care, addressing this failure of healthcare providers during EBOV (1). Health systems need to promptly assess how they could be configured to treat patients or prevent new infections, and then focus on experimental therapies. In the initial stages of a crisis, it may be appropriate to focus the limited resources available on clinical care so as not to detract from the clinical response and incorporate research later when staff is not spread as thin (2).
However, many argue that clinical research (using experimental therapies) and medical care are conflicting activities. Dr. Sacristan, a clinical researcher in Spain, explains this by stating that the purpose of clinical research is to produce generalizable knowledge useful for future patients, while medical care aims to promote the well-being of individual patients (3). This dichotomy manifests an ethical dilemma for physicians acting as both doctors and as researchers. However, both can be seen as two sides of the same coin in the case of a crisis, ideally conducted in tandem (Figure 1). Early on, medical care provides support for experimental therapies undertaken in the future and allows for greater efficacy, with more patients eligible to enter clinical trials.
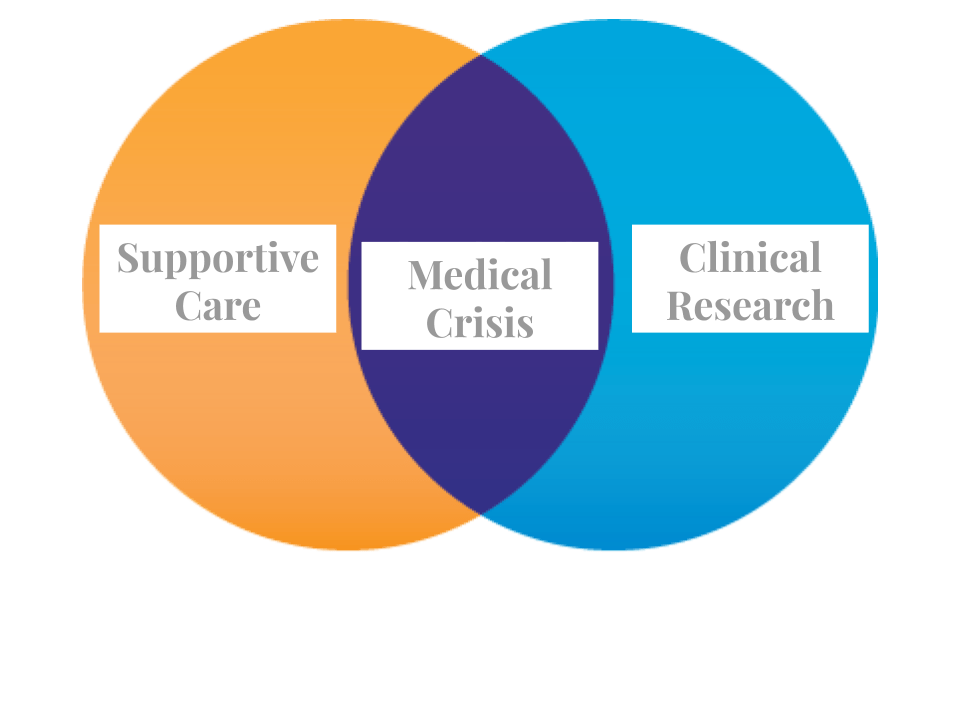
While supportive care is being configured alongside it, there needs to be planning to integrate research for using experimental therapies, because it can take considerable time to obtain the necessary approvals for the study and the type of research being done. First, the possibility of using repurposed drugs needs to be explored. According to the Nature Reviews Drug Discovery October issue repurposing of old drugs is a better path than creating novel therapeutic treatments, given the use of de-risked compounds, with potentially lower overall development costs and shorter development timeline (4).
During EBOV, since early mortality rates were high, some argued that experimental therapies should be given to as many patients as possible, since a number of established drugs were being considered as potential therapeutic agents for the treatment of EBOV (5). The STAC-EE committee on 11-12 November 2014 convened in Geneve for a WHO meeting to decide on the potential experimental products they would be testing (6). The committee believed there needed to be a focus on the availability and predictability of products (brincidofovir and favipiravir), despite no evidence on their effectiveness (7). Others thought that availability was not a reason to study drugs with weak supporting data, favoring drugs with strong supporting preclinical evidence (monoclonal antibodies and small inhibitory RNA), even with less availability (WHO).
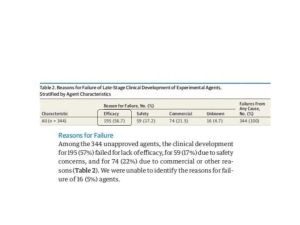
However, during times of crisis, there is little time for these drugs to go through new clinical trials to determine the efficacy of a new patient population. Therefore, assuming there is a strain of time, and supportive care cannot be adequately set up for a majority of patients or the patient has exhausted all available treatment options, patients need to have more say. Proponents of this feel that patients should be able to have access to experimental therapies. This decision is where expanded access, sometimes called “compassionate use”, comes into play. As per the FDA, it allows patients to gain access to an investigational medical product for treatment outside of clinical trials when no comparable or satisfactory alternative therapy options are available (11).
The “Rights and Responsibilities” section of the 1990 Conference on “Expanding Access to Investigational Therapies for HIV Infection and AIDS,” discusses the ethical issues with expanded access. The one extreme argument for freedom of choice, has been that persons infected with HIV have little or nothing to lose, so why limit their access to any drugs? (12). Nevertheless, the problem with this is it fails to recognize the association between desperation and vulnerability (13).
While this is true, totally free access to substances that have little efficacy is different from the opportunity to make logical decisions about products for which there is a reasonable expectation of effectiveness based on preclinical or early clinical data, such as repurposed drugs. Expanded access programs, recognize the right to assume such risks—with the advice and assistance of a personal physician—without abandoning the individual to the forces of the marketplace (14).
In conclusion, experimental therapies need to be administered systemically under the medical community’s guidance in conjunction with supportive care, with a focus on repurposed drugs, and with expanded patient autonomy. However, ultimately, it is the patient’s decision in choosing whether to take part in these therapies.
Citations:
[1] National Academies of Science, Integrating Clinical Research into Epidemic Response: The Ebola Experience. U.S. National Library of Medicine, (2017)
[2] National Academies of Science, Integrating Clinical Research into Epidemic Response: The Ebola Experience. U.S. National Library of Medicine, (2017)
[3] S. José, Clinical Research and Medical Care: towards Effective and Complete Integration. BioMed Central, (2015)
[4] P. Sudeep, et al., Drug Repurposing: Progress, Challenges and Recommendations. Nature Reviews Drug Discovery, (2018).
[5] S. Hussein, et al., Repurposed Therapeutic Agents Targeting the Ebola Virus: A Systematic Review. Elsevier, (2017).
[6] WHO, WHO Meeting of the Scientific and Technical Advisory Committee on Ebola Experimental Interventions – Briefing Note. World Health Organization, (2015).
[7] WHO, WHO Meeting of the Scientific and Technical Advisory Committee on Ebola Experimental Interventions – Briefing Note. World Health Organization, (2015).
[8] F. David, Factors Associated with Clinical Trials That Fail and Opportunities for Improving the Likelihood of Success: A Review. Elsevier, (2018).
[9] National Academies of Science, Integrating Clinical Research into Epidemic Response: The Ebola Experience. U.S. National Library of Medicine, (2017).
[10] National Academies of Science, Integrating Clinical Research into Epidemic Response: The Ebola Experience. U.S. National Library of Medicine, (2017).
[11] FDA, Expanded Access. U.S. Food and Drug Administration, (2020).
[12] National Academies of Science, Expanding Access to Investigational Therapies for HIV Infection and AID. U.S. National Library of Medicine, 19-20 (1991).
[13] National Academies of Science, Expanding Access to Investigational Therapies for HIV Infection and AID. U.S. National Library of Medicine, 19-20 (1991).
[14] National Academies of Science, Expanding Access to Investigational Therapies for HIV Infection and AID. U.S. National Library of Medicine, 19-20 (1991).
Images retrieved from:
[1] https://imgur.com/y7RWDKA
[2] https://imgur.com/6eo1DMp



One thought on “Beyond the Dichotomy: A Systematic Approach to Administering Experimental Therapies”