by Jalwa Afroz ’17
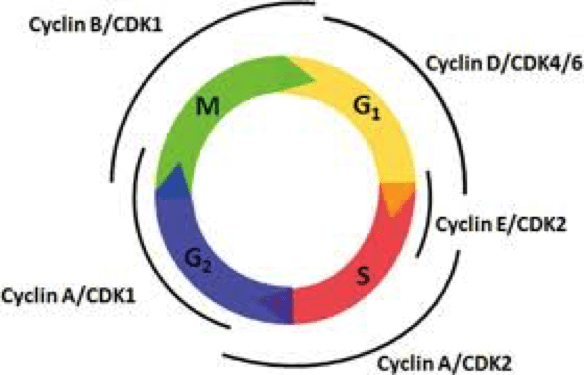
Regulation of the cell cycle is crucial to maintain appropriate cell growth, proper chromosome replication and segregation. Cyclin A2 is one of the many cell cycle regulators that activate cyclin-dependent kinases, Cdk1 and Cdk2, which form Cyclin A2-cdk complexes that activate transcription of DNA replication factors that promote progression through the cell cycle. Conditional cyclin A2 knockout mice revealed the importance of cyclin A2 in cell cycle progression in stem cells and certain neuronal progenitor cells that are important for embryonic growth and differentiation. Since a full knockout of A2 in mice is embryonically lethal, researchers led by Jan van Deursen at Mayo Clinic in Rochester, Minnesota used a combination of knockout, Ccna2, and hypomorphic, Ccna2H, alleles to significantly deregulate cyclin A2 expression without affecting embryogenesis.
Both cyclin A2 deficiency and overexpression have been observed in human tumors. However, it is unknown how cyclin A2 expression contributes to malignant growth of tumors. Deursen and his colleagues treated Ccna2+/+ mice, which had normal functioning cyclin A2, and Ccn2-/H mice, which had reduced levels of cyclin A2, with the carcinogen 7,12-dimethylbenz(a)anthracene (DMBA). Ccna2-/H mice showed a marked increase in tumors in the lung and skin. Also, screenings for chromosomal instability, a characteristic of human malignancies, showed increased aneuploidies (chromosomal segregation errors resulting in an incorrect number of chromosomes) present in Ccna2-/H Mouse embryonic fibroblasts (MEFs). An Eg5 motor protein, or protein that drives centrosome movement at prophase of mitosis, was markedly reduced in Ccna2-/H MEFs, suggesting a catalytic role of cyclin A2-cdk in targeting Eg5 to centrosome microtubules for proper spindle formation and chromosome separation.
The MRN complex, composed of Mre11 mRNA, Rad50, and Nbs1, plays a major role in the DNA replication fork and double-stranded DNA repair. Analysis of cell cycle-dependent expression showed that Mre11 and Rad50 levels peaked, just like Cyclin A2, in Ccna2+/+ MEFs during S and G2 phases. Also, Mre11 and Rad50 levels reduced in Ccna2-/H MEFs. The similar reaction of the MEF cells to either cyclin A2 knockout or Mre11 knockout elucidate a link between cyclin A2 and Mre11 mRNA. This data shows the importance of cyclin A2 in not only cell cycle progression via cdk1 and cdk2 activation and regulation of Eg5 motor proteins important in chromosomal segregation, but also in DNA repair via transcriptional upregulation of Mre11. Previously, cyclins were never implicated in RNA binding. However, this new feature advances the study of the cell cycle and provides potential targets for chemotherapeutics.
References:
- A. Kanakkanthara, et al., Cyclin A2 is an RNA binding protein that controls mre11 mrna translation. Science 353, 1549-1552 (2016). doi: 10.1126/science.aaf7463.
- Image acquired from: http://www.bioscirep.org/content/30/4/24


