By Caleb Sooknanan ’20
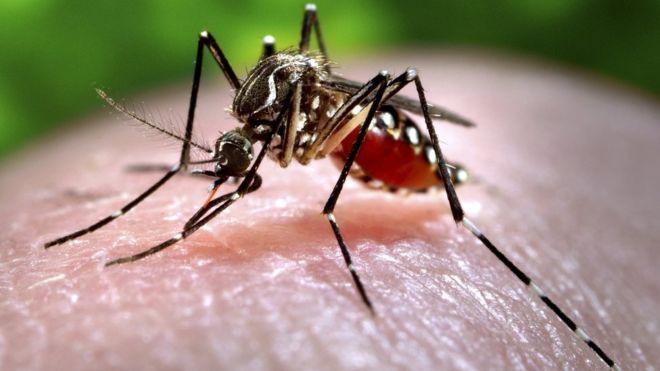
Mosquitoes are known to transmit potent diseases — particularly malaria and dengue fever — among humans. The impact of these diseases is most apparent in sub-Saharan Africa, where over 200 million cases of malaria are reported annually. Many disease control programs in the region have used broad-spectrum insecticides to eliminate mosquitoes and reduce the spread of vector-borne diseases. However, malarial mosquitoes have become increasingly resistant to these insecticides. Doctor Raymond J. St. Leger and researchers from the University of Maryland, Zhejiang University, the University of Queensland, and Institut de Recherche en Sciences de la Santé conducted a study suggesting that a mosquito-killing fungus — genetically engineered to produce spider and scorpion toxins — could combat malarial mosquitoes.
The researchers used the fungus Metarhizium pingshaense, which kills malarial mosquitoes by having its spores germinate and penetrate the mosquitoes’ exoskeletons upon contact. Knowing this, the researchers amplified the power of M. pingshaense by engineering it with genes that expressed neurotoxins from scorpion and spider venom. These toxins would act by blocking calcium, potassium, and sodium channels that are needed for nerve impulse transmission. The researchers then tested the fungal effectiveness against three insecticide-repellant mosquitoes, with two being wild and one being lab-reared.
The engineered strains killed mosquitoes more quickly and efficiently than the conventional fungi. However, the most effective strain utilized two toxins; one was derived from the North African desert scorpion Androctonus australis, while the other was from the Australian Blue Mountains funnel-web spider Hadronyche versuta. The scorpion’s toxin blocked sodium channels, while the spider’s toxin blocked potassium and calcium channels. To ensure that the fungal toxins were not released into their environment, the researchers implemented a specific promoter sequence so that the toxin genes could be activated only by the insects’ blood. This would allow the fungi to infect and selectively kill mosquitoes in Africa.
This study successfully showed that fungi could be genetically altered to serve as biocontrol agents; however, the researchers noted that some female mosquitoes — when infected by fungi — can lay eggs 3 to 4 days after a meal. These mosquitoes could pass their genes onto the next generation, thereby increasing the likelihood of infection. Still, the results demonstrate the need to further understand the effects of fungi on existing mosquito control strategies.
References:
- R. Leger, et al., Improved efficacy of an arthropod toxin expressing fungus against insecticide-resistant malaria-vector mosquitoes. Scientific Reports 7, (2017). doi: 10.1038/s41598-017-03399-0.
- Image retrieved from: http://ichef.bbci.co.uk/news/660/cpsprodpb/DD9F/production/_87953765_c0093043-feeding_mosquito-spl.jpg


