By Rideeta Raquib ’19
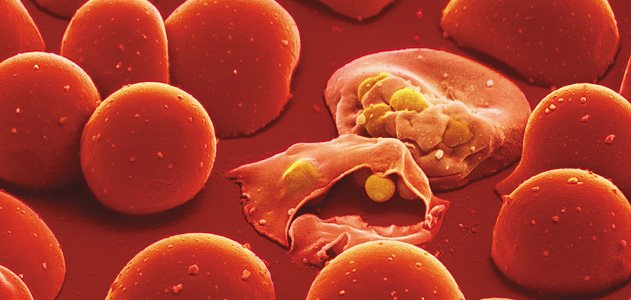
Malaria is a deadly disease caused by single-celled parasites of the genus Plasmodium. Once the body is infected, sporozoites migrate to the liver and replicate to merozoites. A sporozoite in a motile, sporelike phase during a parasite’s asexual reproduction. A merozoite is the growth stage, which occurs in the bloodstream. The toxins released by merozoites destroy red blood cells and can causes severe health complications, such as fevers. The replication of the sporozoites occurs in an intracellular membrane-bound compartment called a parasitophorous vacuole (PV) via schizogeny, which involves the development of micronucleated forms of cells called schizonts. It is during the schizont development that various parasite proteins, such as the serine rich antigen (SERA) family, collect in the PV.
After the parasite goes through multiplication, the infected cells burst. This allows the parasites to escape from the host cell in a process known as egress. Researchers at the Francis Crick Institute and The London School of Hygiene & Tropical Medicine identified a protein called SERA5 involved in the regulation of egress in asexual blood stages of P. falciparum.
In a previous study, the SERA5 gene was deleted utilizing a technique called floxing in two parasite clones. These cells were treated with rapamycin (RAP) that functions in inhibiting cytokine production. Cytokines are substances secreted by the immune system to alert other immune cells of intruders and stimulate an immune response. The schizonts of these clones were analyzed with immunofluorescence (IIFA) at the conclusion of the first erythrocytic cycle, and identified no SERA5-specific signal. The present study expanded upon this finding by comparing the clones with the SERA5 gene deletions and regular cultures. There seemed to a 50 percent reduction in the number of intracellular parasites in the RAP-treated cultures in the cycle after a 44 hour interval. After 12 cycles or 24 days, the cultures without the deletion outgrew the group, but SERA5 parasites were still present in the RAP-treated cultures. This illustrates that the loss of SERA5 expression reduced the rate of parasite replication, but did not terminate it completely. Further examination revealed that the clones with a loss of SERA5 had accelerated schizont rupture but a defective egress.
Overall, this study provides a valuable insight on the mechanisms involved in the replication and growth of P. falciparum. Although cells lacking the SERA5 gene does not completely eliminate the parasites, these findings may inspire future studies to employ conditional techniques in order to isolate malaria mutants that lack expression of specific genes and understanding the functions of the proteins associated with those genes.
References:
- Christine R. Collins, Fiona Hackett, Jonathan Atid, Michele Ser Ying Tan, Michael J. Blackman. The Plasmodium falciparum pseudoprotease SERA5 regulates the kinetics and efficiency of malaria parasite egress from host erythrocytes. PLOS Pathogens, 2017; 13 (7): e1006453 DOI: 10.1371/journal.ppat.1006453
- Image retrieved from: http://www.emedmd.com/sites/default/files/malaria%20parasite.jpg


