Simran Kaur ‘20
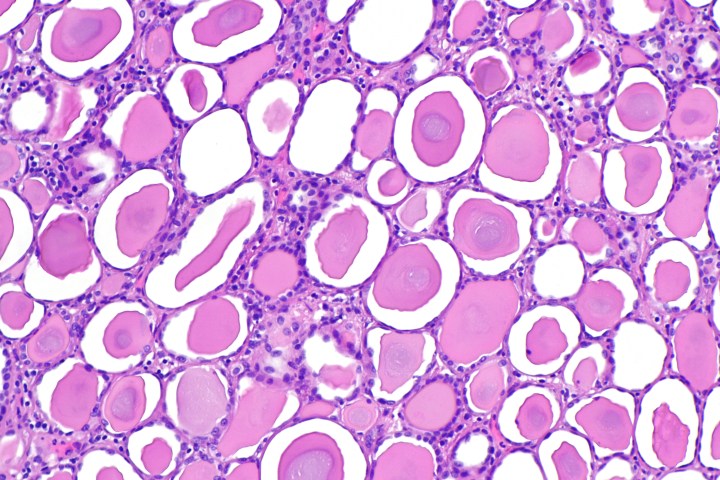
Kidney fibrosis, the accumulation of excess tissue, is the last pathway in end-stage renal failure. Examination of kidneys afflicted with renal disease in both animal and human models has shown a defect in the function of mitochondria. Mitochondria are responsible for the production of energy (ATP) in the cell, funding the processes of toxic waste removal from the blood and the regulation of electrolytes and fluids in the kidneys. Mitochondrial transcription factor A (TFAM) is an important regulator of the gene expression of mitochondria; TFAM is crucial for the transcription, replication, and final structure of mitochondrial DNA. Researchers in this study hypothesized a correlation between kidney function and TFAM in the progression of kidney disease.
To determine the function of TFAM in the development of renal failure, the gene encoding for the expression of TFAM was deleted in the kidney tubules of the crossbred offspring of Ksp-Cre and Tfam mice. The knockout mice, without TFAM, presented significant growth retardation, epithelial atrophy, azotemia (high nitrogen levels in the blood), and interstitial fibrosis. In addition, Sirius red staining showed expression of genes involved in chronic kidney disease, such as Tgfb11, Col1, and Vim in the knockout mice. The gradual tubule atrophy in these mice and profibrotic progression led to end-stage renal failure at just 12 weeks of age, and death in all mice by 16 weeks. Subsequent immunohistological analysis of the mitochondrial proteins in both samples of mice showed that the deletion of TFAM in knockout mice caused a decrease in mtDNA, mitochondrial gene expression, and intracellular ATP levels.
433 dissected human renal tubule samples were collected in this study; RNA sequencing showed that the expression of mitochondrial genes positively correlated with GFR (kidney function). TFAM levels are significantly lower in patients with chronic kidney disease, and therefore it was concluded from both human and mice samples in this study that its levels negatively correlated with the progression of fibrosis in renal failure. Regardless, the observations in this experiment show the need for further studies to determine and understand the relationship between renal mitochondrial metabolism and kidney disease.
References:
- K.W Chung, et al., Mitochondrial damage and activation of the STING pathway lead to renal inflammation and fibrosis. Cell Metabolism, (2019). doi: 10.1016/j.cmet.2019.08.003
- Image retrieved from: https://commons.wikimedia.org/wiki/File:Thyroidization_of_the_kidney_–_intermed_mag.jpg



2 thoughts on “Role of Mitochondrial Gene TFAM in The Progression of Renal Disease”